Melek Umay Tüz Şaşik, Elif Tuğçe Karoğlu Eravşar, Meriç Kınalı, Ayça Ergül Arslan and Michelle Adams have published an article titled "Expression Levels of SMAD Specific E3 Ubiquitin Protein Ligase 2 (Smurf2) and its Interacting Partners Show Region-specific Alterations During Brain Aging".
Abstract
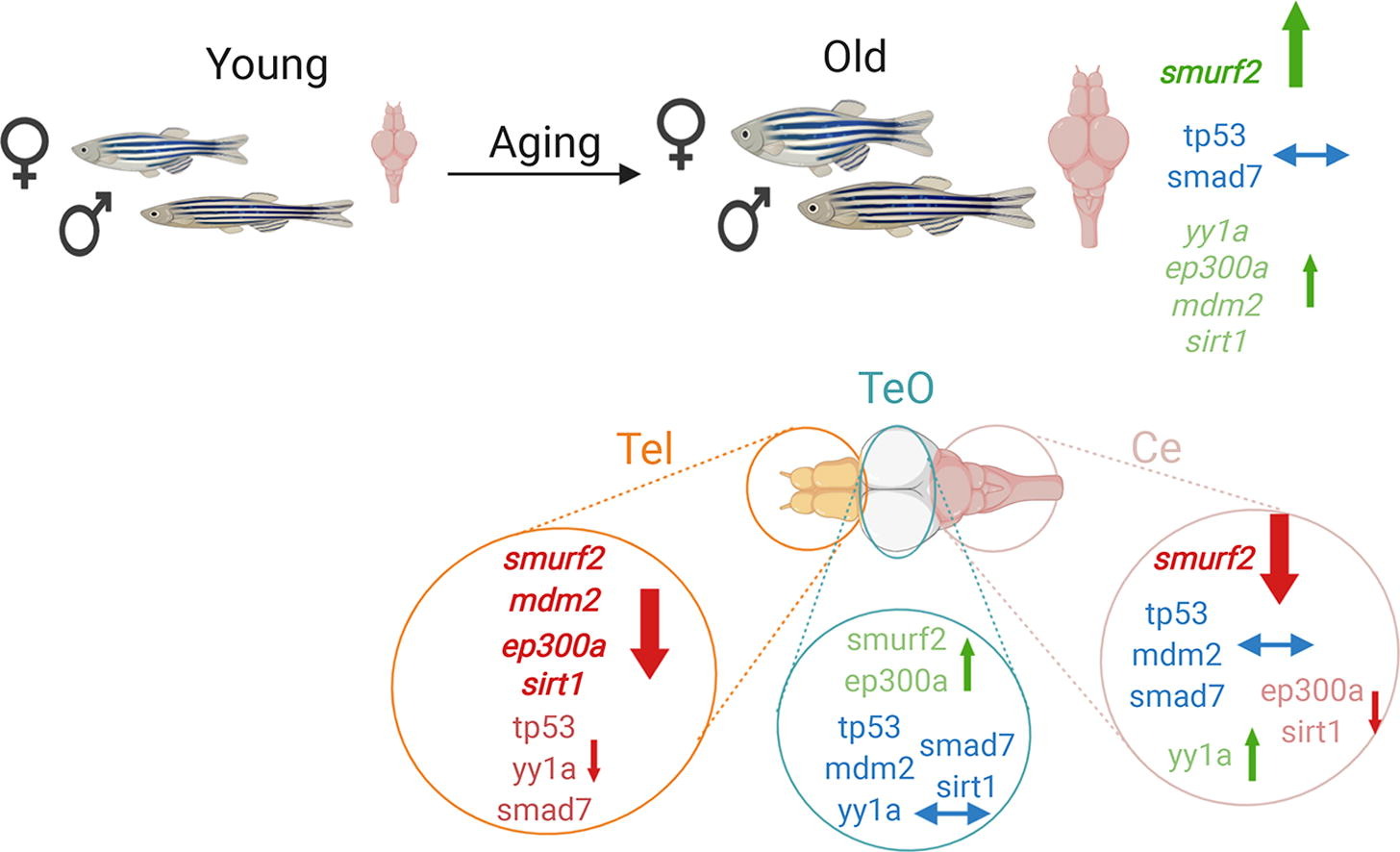
Aging occurs due to a combination of several factors, such as telomere attrition, cellular senescence, and stem cell exhaustion. The telomere attrition-dependent cellular senescence is regulated by increased levels of SMAD specific E3 ubiquitin protein ligase 2 (smurf2). With age smurf2 expression increases and Smurf2 protein interacts with several regulatory proteins including, Smad7, Ep300, Yy1, Sirt1, Mdm2, and Tp53, likely affecting its function related to cellular aging. The current study aimed at analyzing smurf2 expression in the aged brain because of its potential regulatory roles in the cellular aging process. Zebrafish were used because like humans they age gradually and their genome has 70% similarity. In the current study, we demonstrated that smurf2 gene and protein expression levels altered in a region-specific manner during the aging process. Also, in both young and old brains, Smurf2 protein was enriched in the cytosol. These results imply that during aging Smurf2 is regulated by several mechanisms including post-translational modifications (PTMs) and complex formation. Also, the expression levels of its interacting partners defined by the STRING database, tp53, mdm2, ep300a, yy1a, smad7, and sirt1, were analyzed. Multivariate analysis indicated that smurf2, ep300a, and sirt1, whose proteins regulate ubiquitination, acetylation, and deacetylation of target proteins including Smad7 and Tp53, showed age- and brain region-dependent patterns. Our data suggest a likely balance between Smurf2- and Mdm2-mediated ubiquitination, and Ep300a-mediated acetylation/Sirt1-mediated deacetylation, which most possibly affects the functionality of other interacting partners in regulating cellular and synaptic aging and ultimately cognitive dysfunction.